Engility® Trial Management
Clinical trial needs have evolved – isn’t it time CTMS and eTMF did too? Engility® Trial Management is the ideal technology if you’re a business currently using other methods to support your trials or looking for a replacement trial management platform.
With the added benefit of eTMF document storage, there is no need to choose between two disparate systems when one will do.
CTMS is the “brain” and eTMF the “memory” enabling users to know what’s happening in the process at all times using an integrated solution.
Understanding CTMS: Key Fundamentals & Benefits
According to Jason Jones, a CTMS Subject Matter Expert, in today’s fast-paced clinical trial environment, having the right tools is not just beneficial but essential. One of the most vital of these tools is a Clinical Trial Management System (CTMS).
Benefits of Unified Trial Management
Explore the benefits of consolidating CTMS and eTMF. Typically disparate systems now with unified user experience and integrated workflows.
Unified Trial Management
Engility® Trial Management eliminates the need for investing in multiple systems, reducing information fragmentation and the cost and complexity of integrating disparate systems.
Global Collaboration
Engineered for global access with support for multiple languages. This makes it simple to manage users and roles with a flexible security model that can be adapted to meet the needs of each organisation and increase collaboration with partners.
Usability
The intuitive, interactive and engaging user interface provides a seamless, efficient user experience to delight end-users and drives user engagement.
Interoperable
Interoperability with other eClinical applications optimises trial governance and oversight, increasing the visibility and transparency of clinical trial information.
Full Data Lifecycle
Unlike other products, users capture all data within the system without the need for 3rd party tools. This ensures full audit history and the accessibility of all data to users without the risk of loss and burden of reconciliation.
Trial Management Capabilities
Engility® Trial Management increases productivity supporting efficient management of your clinical trials.
Admin & Configuration
Leverage company-wide templates and configuration, with the ability to make study-specific modifications to easily configure your Trial Management requirements from simple things like dropdown values to more complex workflows and access controls.
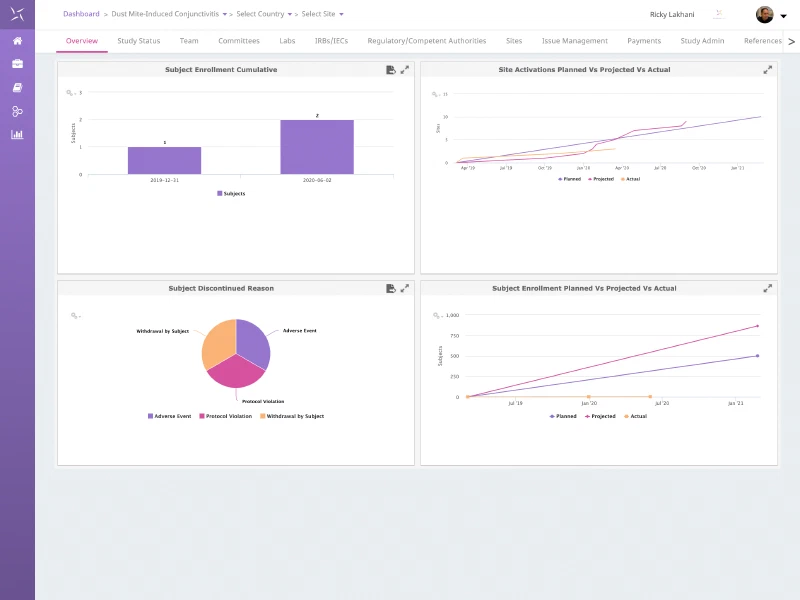
Study & Contact Management
Utilise the global contact management capability of the Engility platform to seamlessly track and manage study and site team members, labs, IRBs/IECs, committees and regulatory authorities for each trial. Leveraging the platform contact database reduces data entry errors and need for reconciliation.
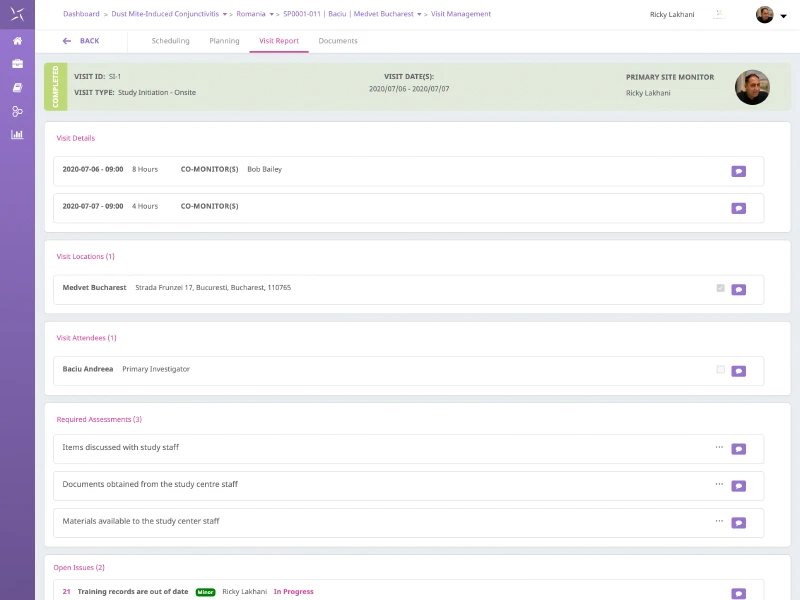
Monitoring
Enable effective oversight of sites with configurable and collaborative site monitoring capability which supports traditional and risk-based approaches. Manage the monitoring plan with visit forecasting, templates and benefit from a smart, intuitive user interface promoting structured data capture and reporting of all monitoring activities.
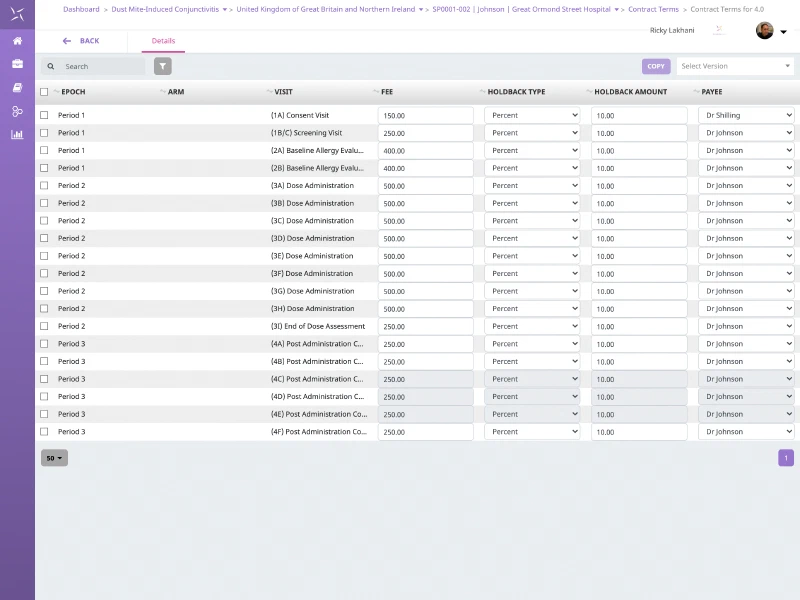
Site Contracts & Payments
Plan, track and support global site and investigator payments including multiple payees. Manage multiple versions of site contract terms including fees and holdbacks. Schedule and trigger payment requests that can be tracked at site and study levels.
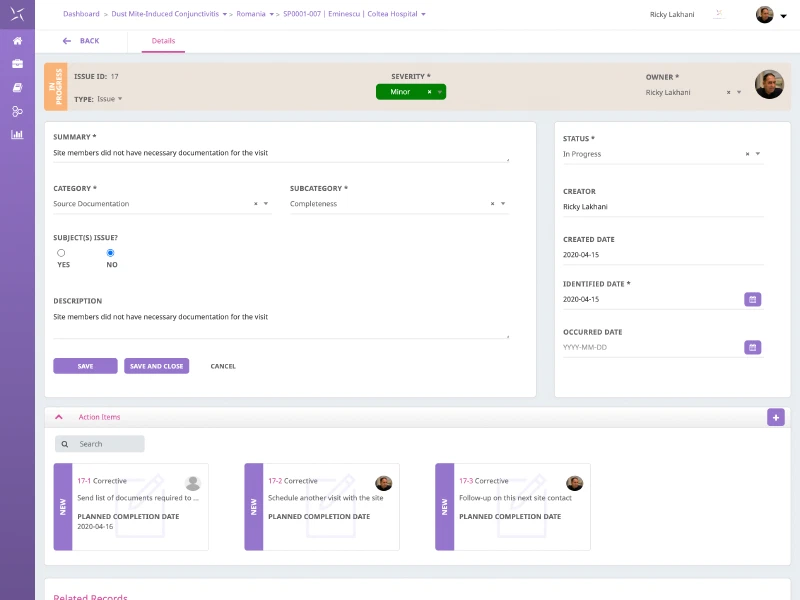
Issue Management
Maintain total oversight and understanding including issues and protocol deviation management centrally. Track and manage issues across the clinical trial lifecycle at multiple levels (study, country, site and subject) with transparent visibility of the issues being found, and the actions being taken to resolve them.
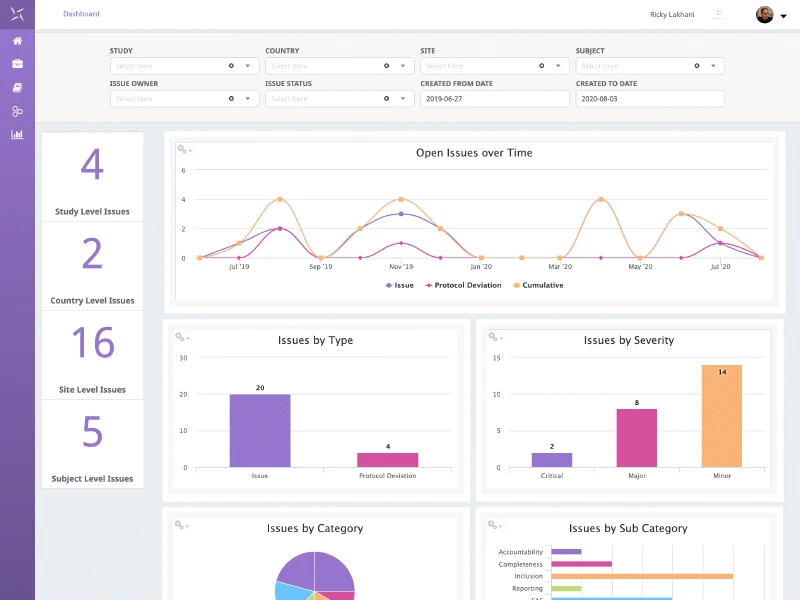
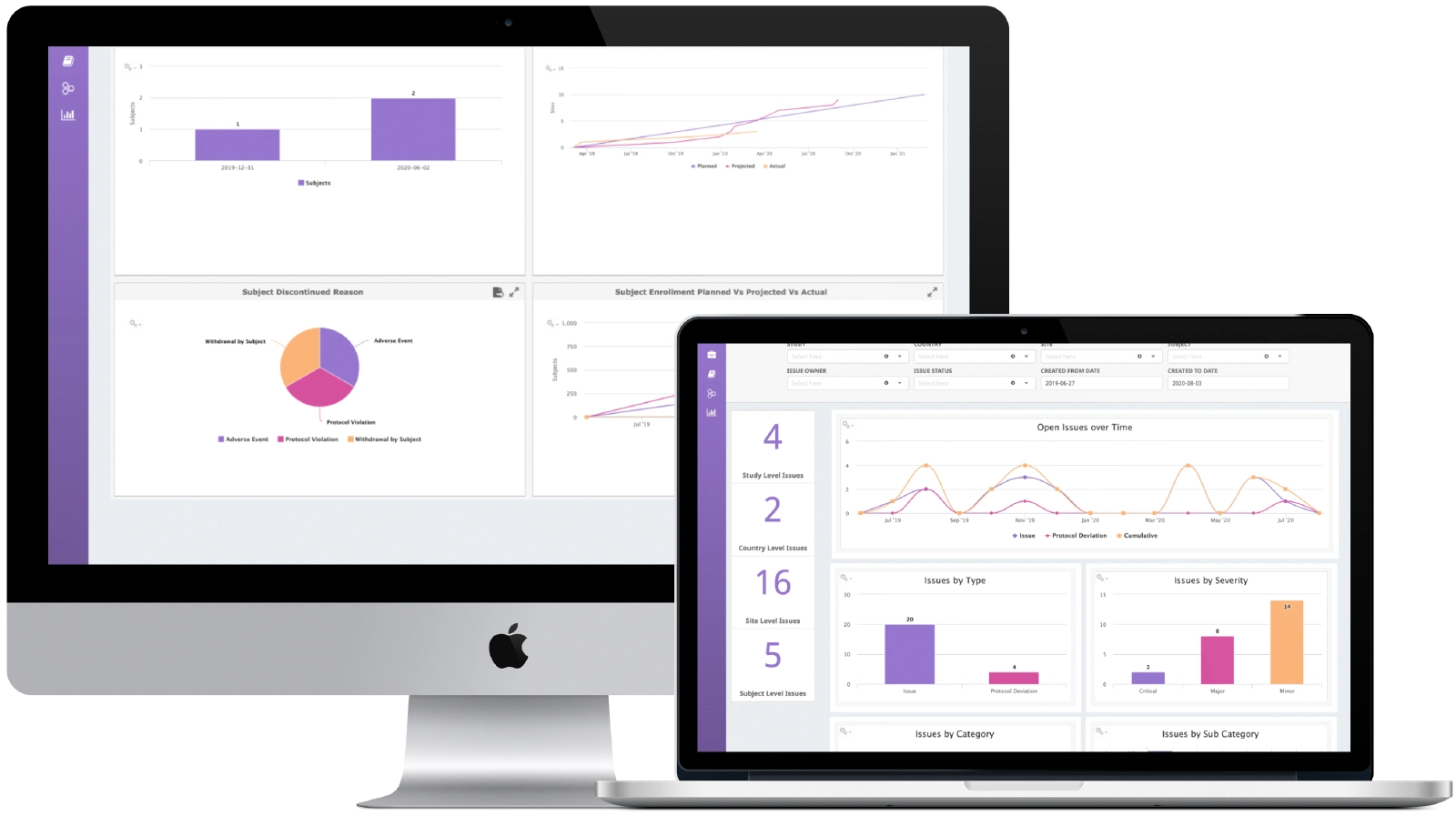
Business Intelligence
Powerful business intelligence through both embedded dashboards and system reports across study management, payments, issue management, TMF and administration. Select and filter reports across study, country and Site. Powerful, searchable, user-friendly audit trail supports regulatory compliance.
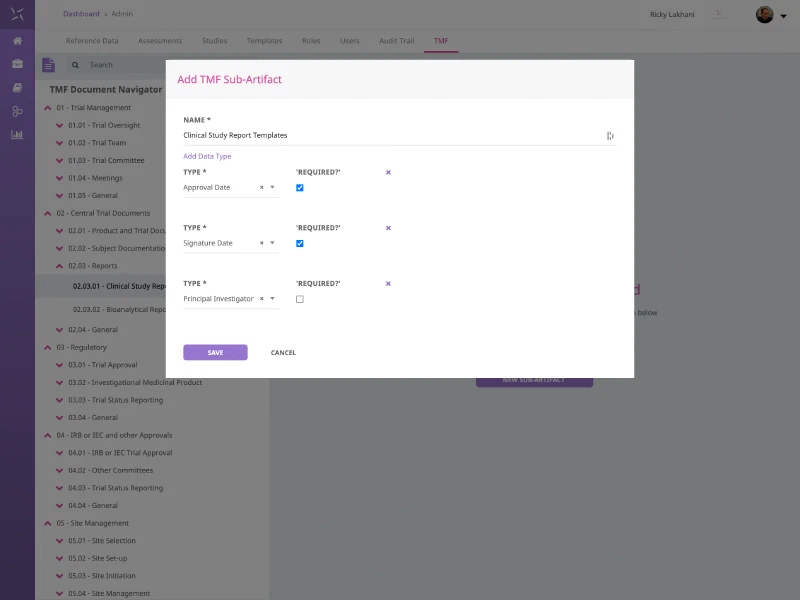
Regulatory Document Tracking
Leverage the DIA TMF Reference Model to track the metadata as well as store hyperlinks to documents being stored in external document repositories.
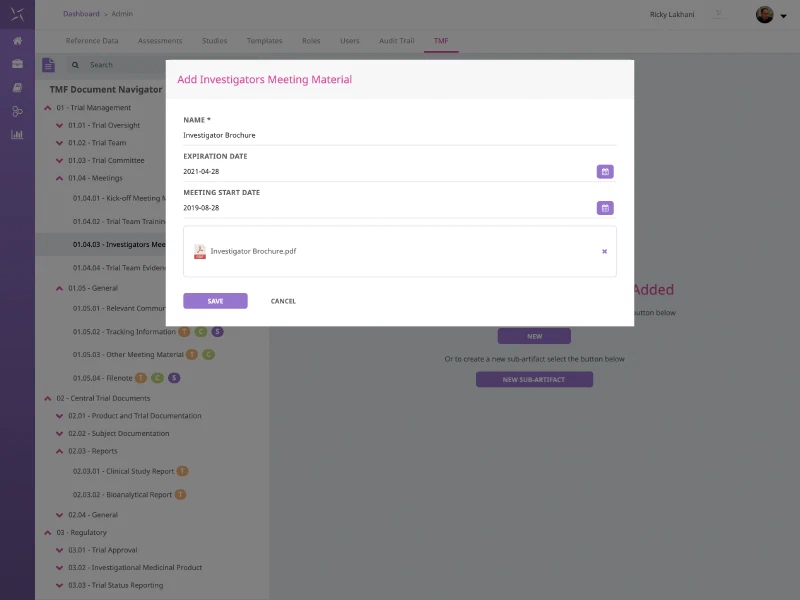
eTMF Document Management
Building on the Regulatory Document Tracking capability, go one further by utilising Engilty® Trial Management to store documents alongside the tracking data in one single, integrated system. Track document expiration, create placeholders for expected documents and benefit from full metadata and document content searching.
Transform your clinical trials
Get in touch to transform your clinical trials using Engility® Trial Management