
Advancing human health together
Helping companies reduce risk and optimise cost through integrated business processes, real time intelligence and smarter management of clinical trials.
Book a demo
Engility®
Intelligent, elegant and efficient trial management
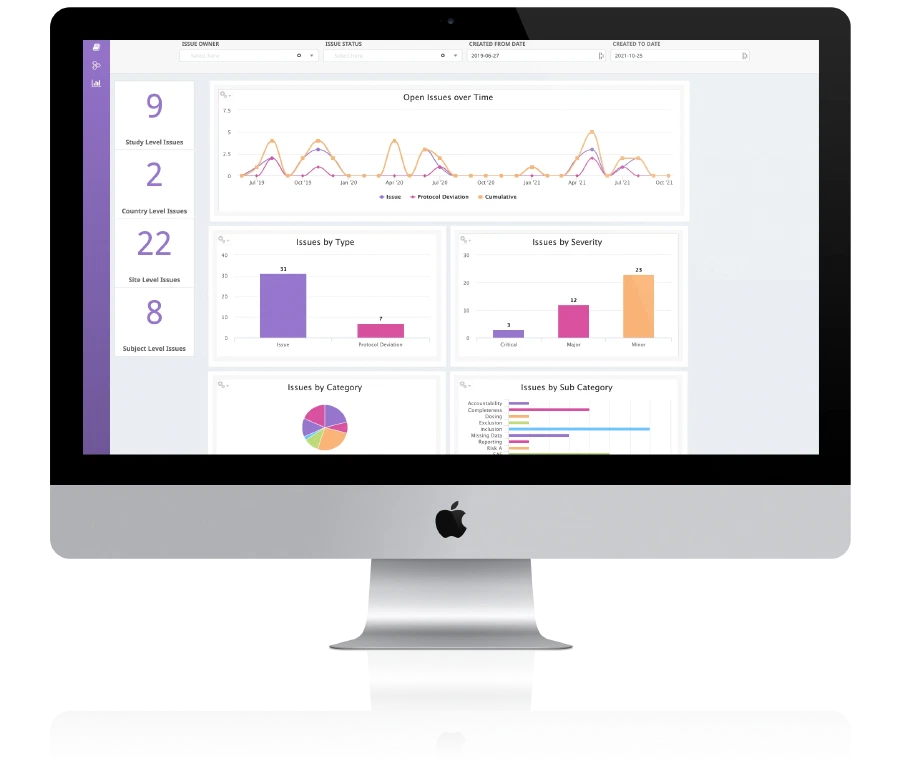
Engility® , an adaptive platform for enabling smarter management and execution of clinical trials. A cloud based flexible trial management platform for companies of all sizes. From single to volume based trials with no user based pricing. See the difference a unified platform makes to your organization.
These features are our differentiators. And it only begins here:
Unified
Single system for trial management and document management ctms + etmf
Supply chain collaboration
Share the system between sponsors and suppliers for greater efficiency and transparency allowing for better decision making and reduced risk
Rapid implementation and configuration
Faster implementation within weeks instead of months
Integration
Includes standardised integration with EDC further reducing cost and timelines We do this better than anyone else.
Watch How Engility Trial Management is Transforming Clinical Trials for Life Science Organizations
Some of our valued customers
Who it’s for and why
Engility has been carefully engineered and designed by clinical technology experts to support a range of different companies including:

Biopharma
Maintain complete trial oversight for internal and outsourced clinical trials.
Medical Device
Oversight and control of Medical Device Regulation (MDR) initiatives

Animal Health
Maintain trial oversight for internal and outsourced clinical trials

Contract Research Organizations
Flexible configuration for different sponsor requirements

Academic Institutions
Affordable enterprise technology reduces total cost of ownership

Nutraceuticals & Cosmetics
Link people, processes, and information via one source of centralized data






















