Engility® CTMS
Smart Technology
Engility CTMS is a SaaS cloud platform built with agility at its core and intelligently engineered to evolve. The progressive platform is designed to rapidly deliver new product updates within a continuous validation framework facilitating accelerated innovation. Flexible and scalable in order to meet the needs of all organizations, Engility CTMS enables data-driven decision-making.
Traditional challenges of poor usability, legacy applications, lengthy implementation cycles, slow innovation, and costly maintenance have been overcome by our creative use of agile flow-based engineering practices.
Whether you are running a few or hundreds of trials, Engility CTMS can support your needs and your specific clinical trial sourcing model. Let Engility CTMS be your first step towards unified trial governance.
Manage Your Clinical Trials Better With Engility CTMS
Product Features
Enterprise Trial Management
In clinical research and healthcare, there are many factors that contribute to the quality of data collected and subsequently analysed that provides evidence. Structured data collection can reduce errors and increase the quality of data captured, ultimately reducing clinical research timelines.
The ability to track study progress including enrolment, milestones and essential documents, maintain organisations and associated contacts including global investigators and manage GCP oversight across flexible supply chains, provides the foundation for a corporate knowledge base. This information hub can be mined to track performance across clinical trials, and organisations can use the metrics to learn from and improve future studies.
Flexible Monitoring
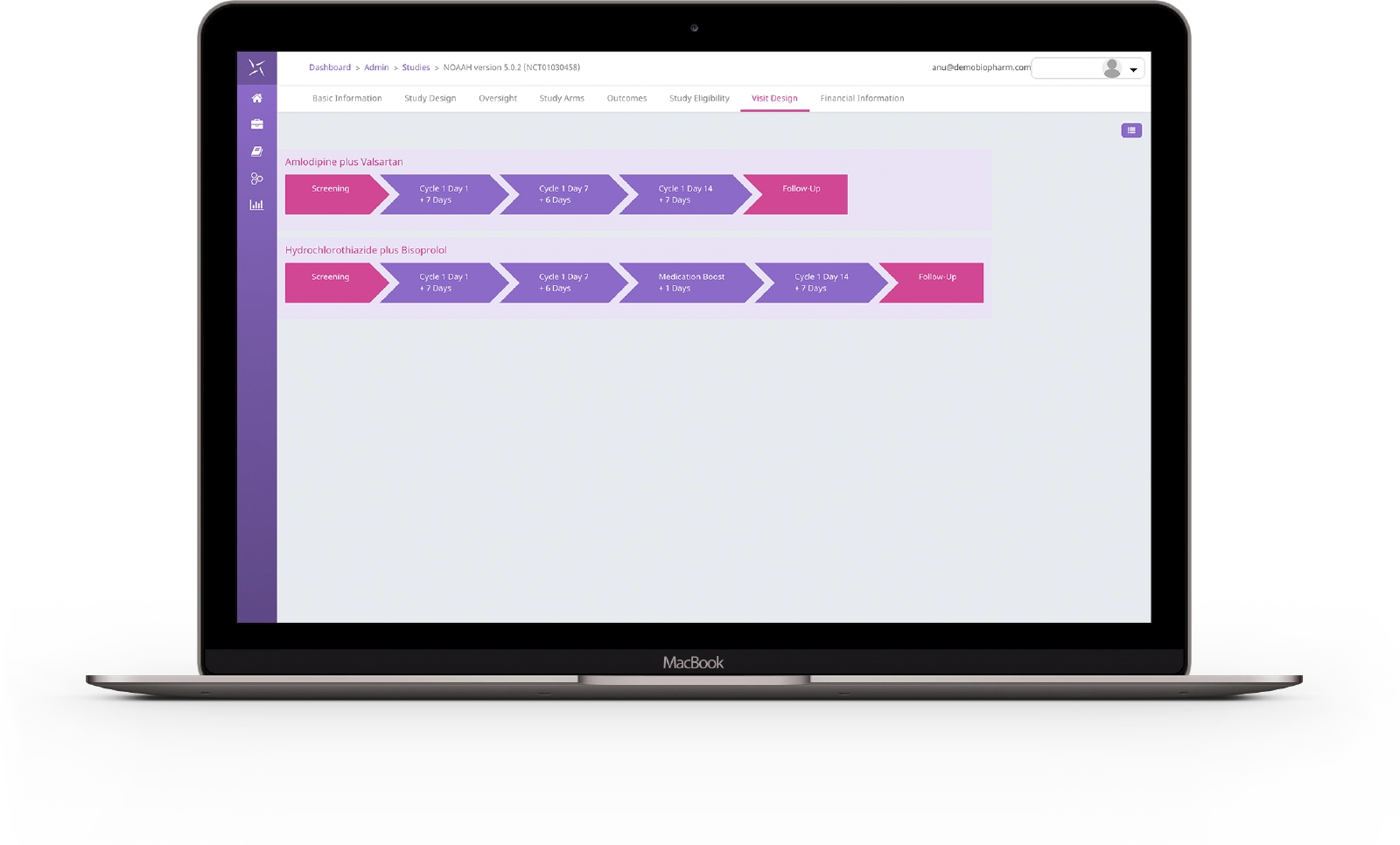
The regulatory landscape has evolved with increased requirements for risk management plans, risk evaluation and minimization strategies. With ICH GCP E6 R2 necessitating a “systematic, prioritized, risk-based approach to monitoring clinical trials” the need for an intuitive monitoring solution has never been greater. With a focus on configurability and collaboration, the EngilityTM CTMS monitoring solution has been designed to address many of the pain points with monitoring today, to enable effective oversight of clinical trials. It provides a workspace to manage all monitoring visits (onsite/remote/telephone) via a simple, perceptive interface to support working in the system.
Engility CTMS presents an intuitive interface to not only configure but visualise monitoring requirements across the study, with support for both traditional and risk-based monitoring schedules allowing for targeted monitoring approaches per site risk. The ability to configure specific monitoring requirements, frequencies and recurrence provides detailed forecasting of monitoring activities on a site by site basis. The flexible interface allows for the creation and management of letter, visit report and addendum templates at customer and study levels. Metadata on templates ensure that only specific templates are available at given points of the workflow, reducing overhead on end users, and ensuring data quality.
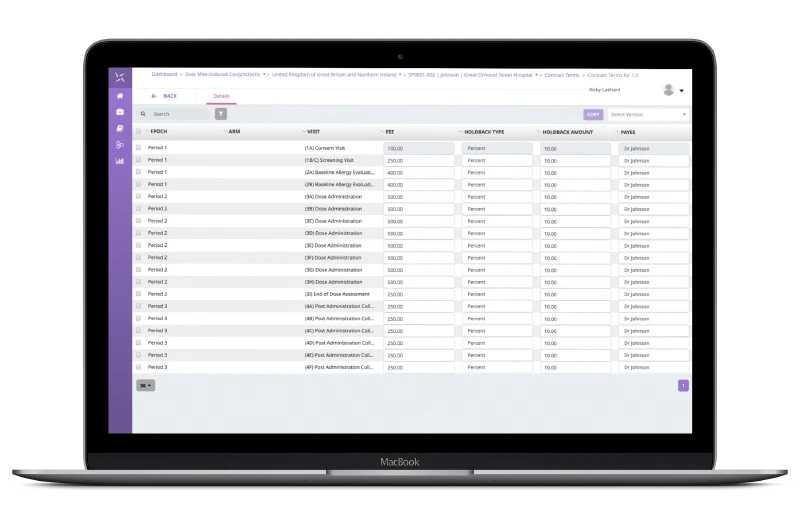
Payments
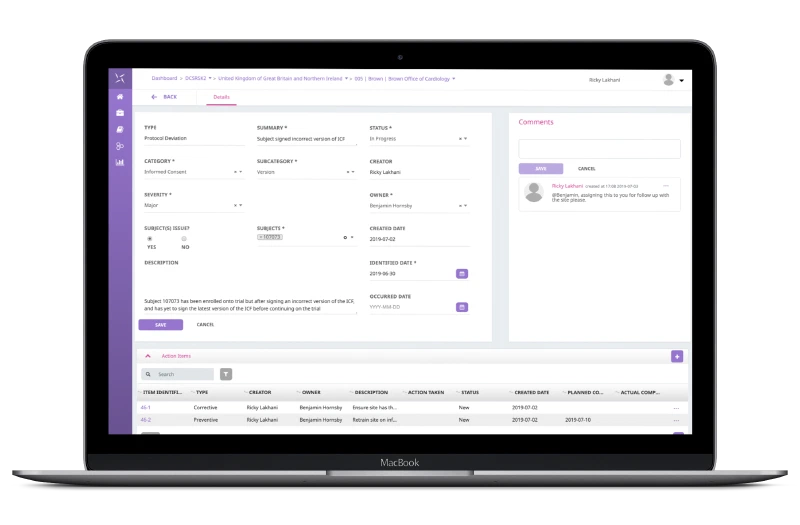
Issue Management
Engility CTMS provides a single, collaborative place to manage all issues (study, country, site, and subject) across the study lifecycle, including protocol deviations. Centralised issue management reduces the need for external manual tools and increases visibility across study teams. Integrated site monitoring capabilities ensure automated documentation of issue management via monitoring visit letters and reports.
With a focus on structured data capture through configurable fields, the quality of data capture is increased, and data entry errors are reduced. Structured data capture provides the ability to analyse and report across different aspects of the issue management spectrum.
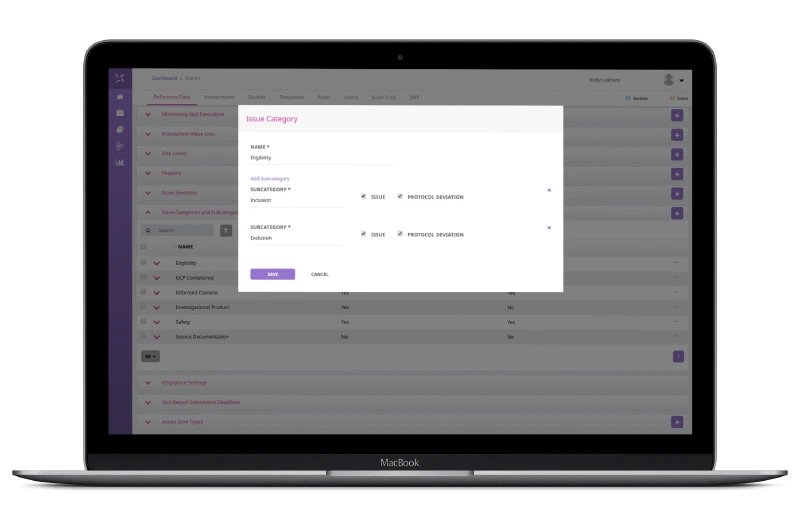
Configurable Design
The globalization of clinical trials poses additional challenges and coordination is required of sponsors to ensure timely communication among all stakeholders in all regions. The globalized access management functionality enables users to easily manage all issues at study, country, site and subject level. Protocol deviations and other study issues can be handled in a central, collaborative, intuitive interface.
The Engility Platform is engineered for global access with support for multiple languages. The enterprise CTMS makes it simple to manage users and roles with a flexible security model that can be adapted to meet the needs of each organisation. It removes the complexity and overheads of managing software, infrastructure and ongoing change allowing you to focus on your clinical trials.
Modern enterprise cloud software supports organisations of all sizes with fast implementation that promotes the sharing of data across the organisation. Our strategy to consolidate data from disparate sources into a single connected platform provides organisations with affordable enterprise control of their clinical trials no matter how many countries or sites are involved. Interoperability with other eClinical applications truly optimises trial governance and oversight, increasing visibility and transparency of clinical trial information. The ability to select and filter reports across study, country and site improves visibility and management of information supporting regulatory compliance.