All / News

PHARMASEAL, a provider of clinical trial management and governance solutions, announced today that it has secured its first Medical Device customer – an award-winning US-based …

A £1 million crowdfunding campaign launched by PHARMASEAL to revolutionise global clinical trials and vaccine development has reached the next campaign milestone. Launched by the …

PHARMASEAL, a new provider of clinical trial management and governance solutions, today announces the launch of a £1 million crowdfunding round with Seedrs. The capital raised will …

PHARMASEAL, a new provider of clinical trial management and governance solutions, announced today that a leading US health outcomes and technology company has selected Engility® …
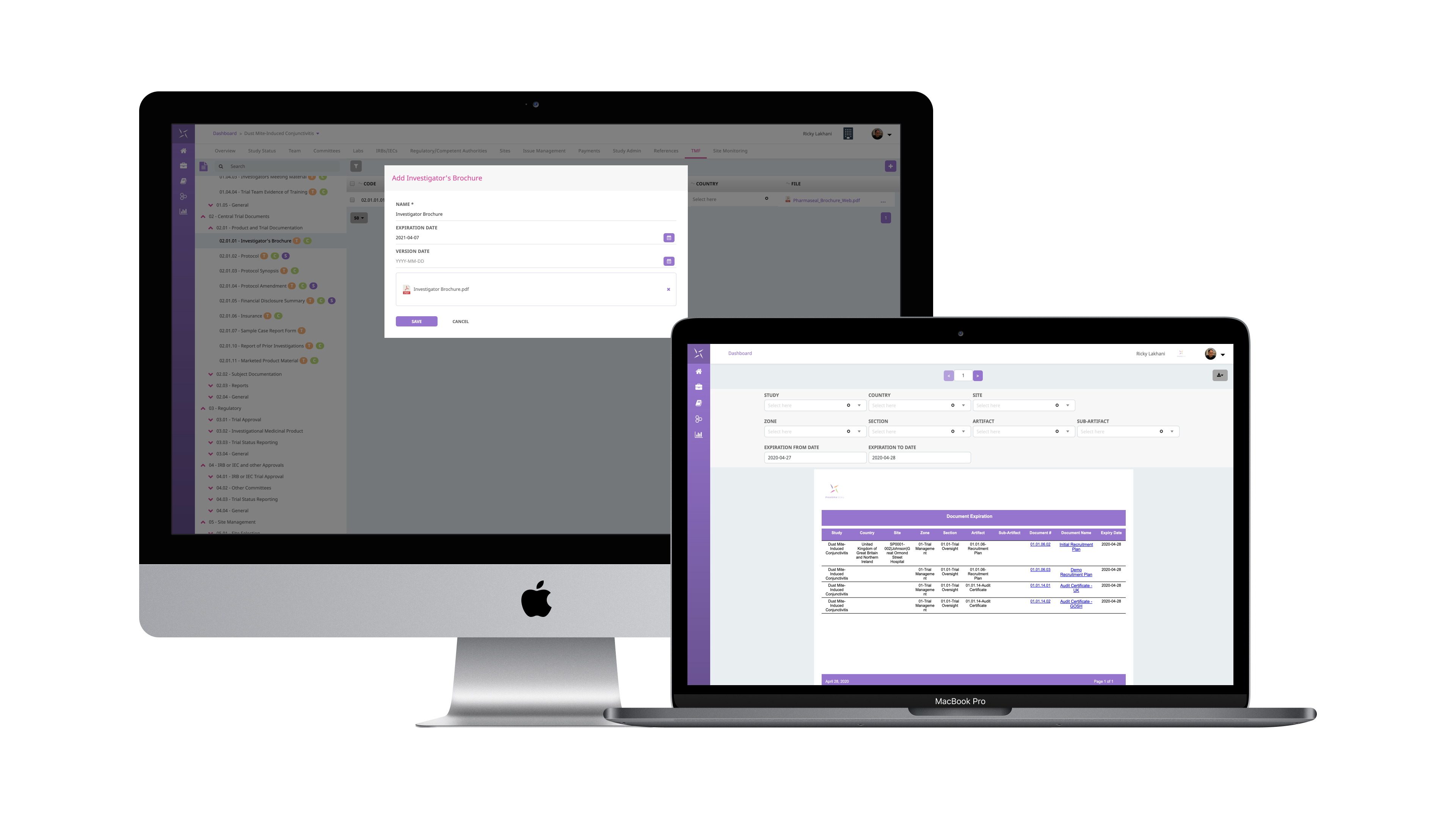
PHARMASEAL, an innovative provider of enterprise trial management solutions expands the capability of the Engility® clinical trial management platform. Electronic trial master file …

Jenni Spinner from Outsourcing Pharma speaks to Daljit Cheema, CEO of PHARMASEAL about the recent offer of no cost CTMS for life science businesses involved with COVID 19 …